Periodic Table GCSE: Complete Guide to Elements, Groups, and Trends
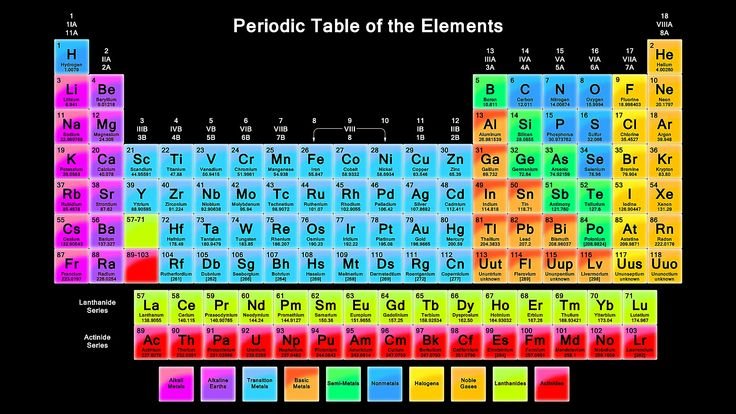
The periodic table GCSE topic is one of the most important foundations in chemistry education. It organizes all known chemical elements in a structured way that helps students understand how matter behaves, reacts, and forms compounds. At GCSE level, mastering the Periodic Table GCSE is essential because it appears in almost every chemistry exam question, from basic element identification to complex reaction predictions. Understanding it properly gives students a strong advantage in both theory and practical science.
The periodic table is not just a list of elements—it is a scientifically designed system that groups elements based on their atomic structure and properties. Each element is arranged according to its atomic number, which represents the number of protons in its nucleus. This arrangement allows patterns and trends to emerge, making it easier to predict how different elements will behave in chemical reactions. For GCSE students, this predictive ability is key to answering exam questions confidently.
Learning the Periodic Table GCSE also helps students understand how the natural world is built. Everything from water and air to metals and gases is made from elements found in the table. By studying it, students begin to see how chemistry connects to real-life applications such as medicine, engineering, and environmental science. This guide will break down the structure, groups, trends, and revision techniques needed to succeed in GCSE chemistry.
Structure of the Periodic Table GCSE
The periodic table is arranged in order of increasing atomic number, meaning each element has one more proton than the previous one. This systematic arrangement is what gives the table its structure and logic. The horizontal rows are called periods, and they represent elements with the same number of electron shells. As you move across a period, elements gradually change from metals to non-metals, showing a clear pattern in chemical behavior.
Vertical columns are known as groups, and elements within the same group share similar chemical properties. This is because they have the same number of electrons in their outer shell, which determines how they react with other elements. For example, all elements in Group 1 have one outer electron and are highly reactive metals. Understanding groups and periods is essential for answering GCSE questions about trends and properties.
Atomic structure plays a major role in how the Periodic Table GCSE is organized. Every element consists of protons, neutrons, and electrons. The atomic number represents the number of protons, which defines the element itself. Meanwhile, the arrangement of electrons around the nucleus determines chemical behavior. GCSE students are expected to understand how electron configuration links to an element’s position in the table.
Element symbols are also a key part of GCSE chemistry. Each element has a unique one- or two-letter symbol, such as H for hydrogen or Na for sodium. Memorizing common symbols is important for exams because questions often require quick identification of elements and their properties.
Groups in the Periodic Table GCSE Focus
One of the most important parts of the periodic table GCSE syllabus is understanding element groups. Group 1 elements, known as alkali metals, are extremely reactive and include lithium, sodium, and potassium. Their reactivity increases as you move down the group because outer electrons become easier to lose. These metals react strongly with water, producing hydrogen gas and alkaline solutions, which is why they are called alkali metals.
Group 7 elements are known as halogens and include fluorine, chlorine, bromine, and iodine. These are non-metals and are also highly reactive, but their reactivity decreases as you move down the group. Halogens react with metals to form salts, such as sodium chloride. Their physical states also change down the group, from gases to liquids to solids.
Group 0, also called noble gases, includes helium, neon, and argon. These elements are extremely stable because they have full outer electron shells. As a result, they are chemically inert and do not easily react with other elements. Noble gases are commonly used in lighting, balloons, and insulation due to their stability.
Transition metals are found in the central block of the Periodic Table GCSE. They have unique properties such as high melting points, strength, and the ability to form colored compounds. Unlike main group elements, they can form multiple oxidation states, making them useful in industrial applications like catalysts and manufacturing processes.
Periodic Trends and Patterns
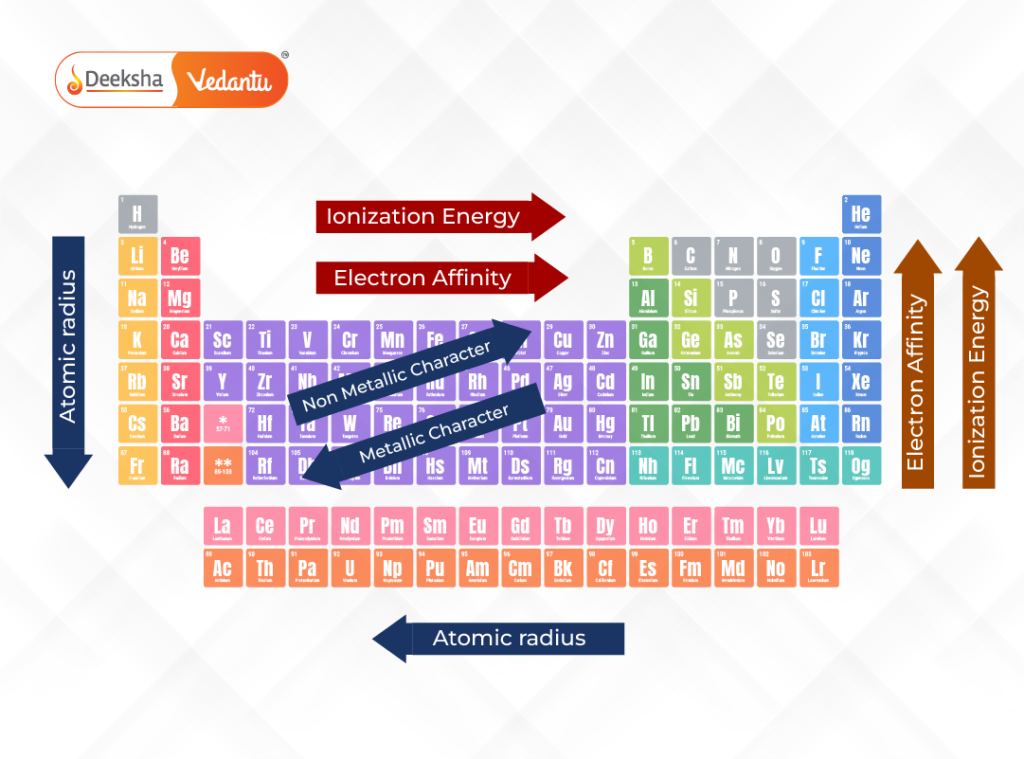
Understanding trends is a crucial part of mastering the periodic table GCSE topic. One of the key trends is reactivity. In Group 1 metals, reactivity increases as you move down the group, while in Group 7 halogens, reactivity decreases. These patterns are directly linked to electron structure and how easily atoms gain or lose electrons.
Atomic size, also known as atomic radius, is another important trend. As you move across a period from left to right, atomic size decreases because the number of protons increases, pulling electrons closer to the nucleus. As you move down a group, atomic size increases because new electron shells are added.
Melting and boiling points also show clear patterns. Metals generally have high melting points due to strong metallic bonding, while non-metals often have lower melting points. However, there are exceptions, and GCSE students are often asked to explain these variations based on bonding and structure.
Metallic and non-metallic character also changes across the table. Metals are typically found on the left side, while non-metals are on the right. Elements in between, known as metalloids, show properties of both. These trends help students predict how elements will behave in chemical reactions.
How to Use the Periodic Table GCSE Exams
In GCSE exams, students are often required to interpret information directly from the Periodic Table GCSE. This includes identifying atomic numbers, understanding group positions, and predicting chemical behavior. Being able to quickly locate elements and understand their properties is essential for success.
Common exam questions include explaining trends, predicting reactions, and describing atomic structure. For example, students may be asked why sodium is more reactive than lithium or how chlorine behaves in displacement reactions. These questions test both memory and understanding of periodic trends.
Effective revision techniques include using flashcards, mnemonics, and repeated practice with past exam papers. Visual learning tools such as color-coded Periodic Table GCSE can also help reinforce memory. Consistent revision is key to mastering this topic.
Importance of the Periodic Table GCSE Chemistry
The Periodic Table GCSE is the foundation of chemistry because it explains how all elements interact. It allows scientists and students to predict chemical reactions, understand bonding, and explore the properties of matter. Without it, chemistry would be far more complex and less organized.
In real life, the periodic table is used in medicine, engineering, environmental science, and technology. From designing new materials to understanding biological processes, it plays a crucial role in scientific advancement. For GCSE students, it is the starting point for more advanced scientific studies.
Conclusion
The periodic table GCSE topic is essential for understanding chemistry at both academic and real-world levels. It provides a structured way to learn about elements, their properties, and how they interact. By mastering its structure, groups, and trends, students can significantly improve their exam performance and scientific understanding.
With consistent practice and a clear understanding of patterns, the periodic table becomes much easier to use and remember. It is not just a chart—it is the key to unlocking the entire subject of chemistry.
FAQs – Periodic Table GCSE
What is the periodic table in GCSE Chemistry?
It is a structured chart that organizes all chemical elements by atomic number and properties.
How many groups are in the periodic table?
There are 18 groups in total.
Why is the periodic table important?
It helps predict chemical reactions and understand element properties.
What are the most important groups for GCSE?
Groups 1, 7, and 0 are most commonly studied.
How can I learn the periodic table easily?
Using repetition, flashcards, and visual memory techniques helps greatly.
What are periodic trends?
They are patterns in properties like reactivity, atomic size, and melting points across the table.
You May Also Read: Stranger Things Lego




